It is important to measure and assess service provision to ensure it is efficient and meeting patients’ needs. Kirsty Mahoney, Wound Care People Ltd, outlines how you can perform clinical audit in your setting to help develop your service.

Reflect on your own practice
Is there a clinical guideline or standard where an audit could assist to identify potential variations in practice and where data collected could be used to improve patient outcomes?
| Aspect | Service evaluation | Clinical audit | Research |
| Purpose | To assess how well a service or care delivery is working in its current form | To measure current practice against agreed standards and identify areas for improvement | To generate new, generalisable knowledge or test a hypothesis or understand cause and effect |
| Main question | What is happening in this service? | Are we doing what we should be doing, according to standards? | What is the best thing to do for this population? May compare interventions or seek to understand people’s perceptions and behaviours |
| Change intent | May identify areas for improvement but does not necessarily involve change | Aims to improve practice and patient outcomes through measurable change | Aims to establish new practices or interventions that could change future care |
| Standards used | No pre-defined standards – looks at current practice or outcomes | Based on explicit standards or guidelines | No pre-defined standards – uses a research question or hypothesis |
| Methods | Data collection to describe or monitor service delivery | Data collection to compare current practice with standards followed by re-audit | Experimental or observational design using rigorous scientific methodology |
| Ethical approval | Not usually required (if no change to patient care) | Not usually required (as care is not altered) but depends on the subject of the audit | Required from a Research Ethics Committee before starting |
| Outcome | Provides information for service planning or management decisions | Lead to specific quality improvement and reassessment of care | Provides new evidence that can inform future guidelines, standards or treatments |
| Example in urology or continence | Reviewing waiting times for continence clinic referrals | Auditing compliance with aseptic technique during catheter insertion | Testing a new catheter coating to reduce infection rates |
Reflect on your own practice?
Have you been involved in any local or national audits in your place of work?
Did you know the outcomes of the audit, and did it change practice?
If you have not been involved directly in any audits, try and find out if any audits have been completed in your area of clinical practice and what were the results?
Reflect on your own practice?
Do you know your organisation’s process for submitting an audit project – if not try and find out by speaking to your local audit or research and development department.
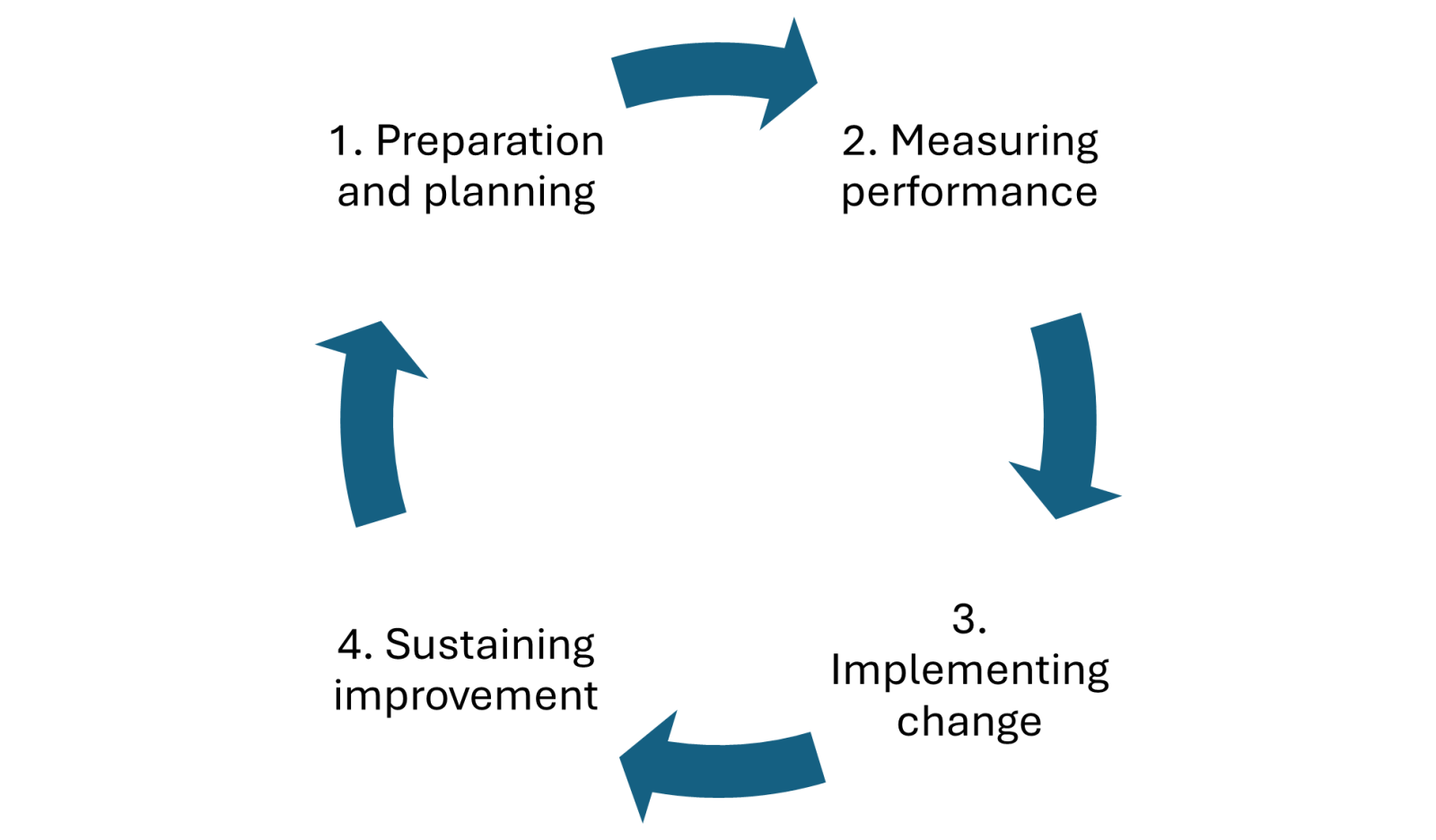
| Set objectives | Why is there a need for the audit and what will it set out to achieve |
| Consider your population – decide how many and who will be included in the audit, e.g. all adult patients on ward xx who had a catheter inserted during the first week of October | You may not be able to audit every patient with the specific condition or procedure (which would be the ideal). In this situation, selecting a sample population may make audit more manageable, but the sample used must be representative of the population as a whole |
| Ethical consideration and data protection | Does not usually need ethical consideration but has to fit within organisational protocols. Data should be collected and stored in line with organisational data protection policies |
| What should we look for to tell us we are providing quality care | Look for evidence-based standards or local policy to measure current practice against, e.g. to prevent infection following catheter insertion standards set out in the National Institute for Health and Care Excellence (2014) guidelines |
| How often should each part of the standard be followed to meet the expected quality? | Target % to be achieved within the audit. Hughes (2012) indicates that targets should aim to achieve 100% compliance |
| Exceptions to the standard | Are there any instances when deviation from the standard would be acceptable? This would have to be clearly identified within the audit |
| Define how and what data will be collected and how it will be safety stored in line with data protection | Clearly define instructions on standards to be measured and how data will be collected, stored and where it will be obtained from (e.g. patient catheter bundle documentation) |
|
